Peritumoral stroma and systemic inflammatory response in cervical cancer
Peritumorózní stroma a systémová zánětlivá odpověď u karcinomu děložního hrdla
Cíl: Srovnání cervikálního stromatu u pacientek s pokročilými stadii karcinomu děložního hrdla s kontrolní skupinou: v období před léčbou srovnat hematologické parametry u pacientek se stejnými parametry v kontrolní skupině; ověřit souvislost stromálních markerů s s prognostickými faktory karcinomu děložního hrdla. Materiál a metody: Prospektivně bylo hodnoceno 16 pacientek s diagnózou pokročilého invazivního karcinomu děložního hrdla. Kontrolní skupinu tvořilo 22 pacientek (s děložním myomem). Bylo provedeno imunohistochemické vyšetření k detekci hladkosvalového aktinu alfa (SMA – alpha-smooth muscle actin) a fibroblasty aktivujícího proteinu alfa (FAP – fibroblast activation protein alpha). Výsledky imunohistochemického vyšetření a hematologické parametry byly vyhodnoceny pomocí Fisherova exaktního testu a Mann-Whitneyho testu. Výsledky: Silné imunobarvení FAP bylo častější u pacientek s karcinomem děložního hrdla v porovnání s pacientkami s děložním myomem (p = 0,0002). Co se týká SMA, silné imunobarvení bylo také ve větší míře zjištěno ve skupině pacientek s karcinomem oproti kontrolní skupině (p < 0,00001). Poměr neutrofily/lymfocyty (NLR) byl vyšší u pacientek s neoplazií děložního hrdla ve srovnání s kontrolní skupinou (p = 0,0019). Souvislost mezi zkoumanými parametry a prognostickými faktory nebyla prokázána. Závěr: Silné imunohistochemické barvení FAP a SMA je ve srovnání s kontrolní skupinou častější u pacientek s karcinomem děložního hrdla. Poměr NLR u nich byl rovněž vyšší.
Klíčová slova:
karcinom děložního hrdla – krevní obraz – hladkosvalový aktin alfa – fibroblasty aktivující protein alfa
Authors:
S. Santana Otano 1
; A. Mendes Melo 2; B . Ferreira Souza 2
; J. Souto Morlin 2
; E. de Castro Côbo 3
; A. C. Macêdo Barcelos 2
; A. Misson; A. Misson Rua Micheletti 3
; Millena Prata Jammal 2
; Eddie Fernando Candido Murta 2
; Rosekeila Simões Nomelini 2,4
Authors‘ workplace:
Service of Radiotherapy, Federal University of Triângulo Mineiro, Uberaba, Brazil
1; Laboratory of Applied Sciences for Women (LaCam), Department of Gynecology and Obstetrics, Federal University of Triângulo Mineiro, Uberaba, Brazil
2; Discipline of Special Pathology, Federal University of Triângulo Mineiro, Uberaba, Brazil
3; Graduate Program in Gynecology and Obstetrics of the Ribeirão Preto Medical School of the University of São Paulo, Brazil
4
Published in:
Ceska Gynekol 2024; 89(2): 95-101
Category:
Original Article
doi:
https://doi.org/10.48095/cccg202495
Overview
Objective: To compare cervical stroma in advanced cervical cancer with the control group; to compare, in the pre-treatment period, hemogram parameters in patients with advanced cervical cancer with the same parameters as the control group; and to verify if there is an association of stromal markers with prognostic factors in cervical cancer. Materials and methods: We prospectively evaluated 16 patients diagnosed with advanced invasive cervical cancer. A control group of 22 patients was used (uterine leiomyoma). Immunohistochemistry was performed to verify the stromal immunostaining of alpha-smooth muscle actin (SMA) and fibroblast activation protein alpha (FAP). Immunostainings and hemogram parameters were compared using Fisher's exact and Mann-Whitney Test, respectively. Results: Strong FAP immunostaining was more frequent in patients with cervical cancer when compared with patients with leiomyoma (P = 0.0002). Regarding SMA, strong immunostaining was also found more in the group of cancer patients compared to the control group (P < 0.00001). The neutrophil-lymphocyte ratio (NLR) values were higher in the cancer patient group compared to the control group (P = 0.0019). There was no association of the parameters studied with prognostic factors. Conclusions: Strong FAP and SMA immunostaining was found more in patients with cervical cancer when compared to the control group. NLR values were also higher in cervical cancer.
Keywords:
uterine cervical neoplasms – alpha-smooth muscle actin – fibroblast activation protein alpha – blood cell count
Introduction
Currently, cancer is the main public health problem in the world and is among the four main causes of premature death in most countries. The incidence and mortality from this disease have been increasing due to aging, population growth, as well as changes in the distribution and prevalence of risk factors, especially those associated with socioeconomic development [1].
The tumor stroma has several cells, in addition to the extracellular matrix that can suppress the host’s immune responses against tumor cells. Through the secretion of immunosuppressive cytokines, metabolic changes and other mechanisms, the tumor stroma provides a complex network of safeguards for tumor proliferation [2].
The tumor microenvironment is composed of a complex network of extracellular matrix and cells such as cancer-associated fibroblasts (CAFs), blood vessel associated smooth muscle cells, pericytes, endothelial cells, mesenchymal stem cells and a variety of immune cells. Changes in the homeostasis of this environment contribute to tumor growth, angiogenesis, metastasis, immune suppression and resistance to treatments [3].
Studies suggest that two stromal markers, alpha-smooth muscle actin (SMA) and fibroblast activation protein alpha (FAP), play an important role in the stroma of a malignant ovarian neoplasm. Stronger FAP immunostaining was found in ovarian cancer compared with benign ovarian neoplasms. In ovarian cancer, immunostaining plus FAP was higher in moderately differentiated and undifferentiated ovarian carcinomas compared to well-differentiated tumors, demonstrating that FAP may play a role in the prognostic prognosis. SMA is expressed in smooth muscle cells of the vasculature and it is the most widely used marker for identifying myofibroblasts. In an immunohistochemical study of ovarian neoplasms, detection of SMA alone has not consistently provided significant results [4,5].
The objectives of the study were to compare the cervical stroma in patients with advanced cervical cancer with the control group; to compare, in the pre-treatment period, blood count parameters (platelets, NLR – neutrophil-lymphocyte ratio, PLR – platelet-lymphocyte ratio, RDW – red cell distribution width) in patients with advanced cervical cancer (stage ≥ IIB) with the same parameters as the control group; and in cervical cancer, to verify if there is an association of immunostaining of stromal markers with pathological (histological type, histological grade and staging) and systemic (platelets, NLR, PLR and RDW) prognostic factors.

Materials and methods
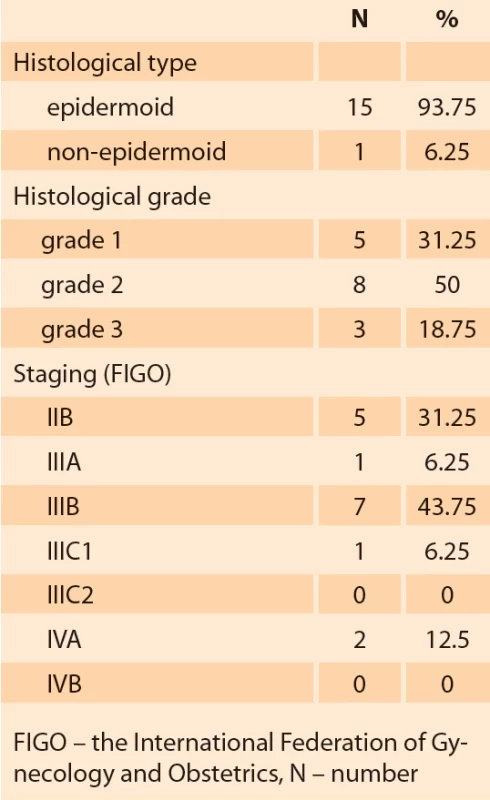
We prospectively evaluated 16 patients diagnosed with advanced invasive cervical carcinoma staging ≥ IIB – FIGO (the International Federation of Gynecology and Obstetrics) [6], confirmed by biopsy, attended from 2020 to 2022. Clinical data, oncotic cytology, blood count parameters (neutrophils, lymphocytes, platelets, RDW) and biopsy data were recorded in a specific database for the study. A control group of 22 patients was used.
The inclusion criterion was women with invasive cervical cancer staging ≥ IIB. Exclusion criteria were pregnancy, other diseases that lead to immunosuppression, use of immunosuppressive drugs and relapse.
Data were obtained from the patients’ medical records. All patients with a Pap smear showing invasive cervical carcinoma underwent cervical biopsy. Staging was performed through clinical and imaging exams. Patients with a stage ≥ IIB were selected for the study. The control group consisted of 22 patients who underwent hysterectomy for uterine leiomyoma without alterations (neoplasia or dysplasia) in the Pap smear prior to surgery and without cervical involvement of the myoma.
Calculation of the NLR was performed by dividing the absolute number of neutrophils by the absolute number of lymphocytes. The division of platelet count and lymphocyte count resulted in the PLR.
Immunohistochemistry was performed to verify stromal immunostaining of SMA and FAP in cervical cancer and the control group.
Consent was obtained from all patients included in the study and from all patients in the control group.
Cervical biopsy collection
Cervical biopsies were stored in formaldehyde and sent to the Special Pathology Department. Biopsy collection is already a routine procedure for the group of patients with cervical cancer. For the control group, the biopsy was performed immediately after the removal of the surgical specimen (uterus), avoiding discomfort for the patient (since a cervical biopsy is not necessary in patients with uterine leiomyoma).
Immunohistochemistry study
Specimens obtained by surgical resection were processed in paraffin and reviewed by an experienced pathologist. We used the streptoavidin-biotin-peroxidase technique. Immunohistochemical staining was performed with the Novolink™ Polymer Detection System (Leica Biosystems, Buffalo Grove, IL, USA). The specific primary antibodies used were anti-aSMA and anti-FAP, according to the manufacturer’s indications.
Two observers evaluated the slides. Intensity of immunostaining in the stroma was subjectively assessed using 0 to 3:
- 0 (no staining);
- 1 (weak staining);
- 2 (moderate staining);
- 3 (strong staining).
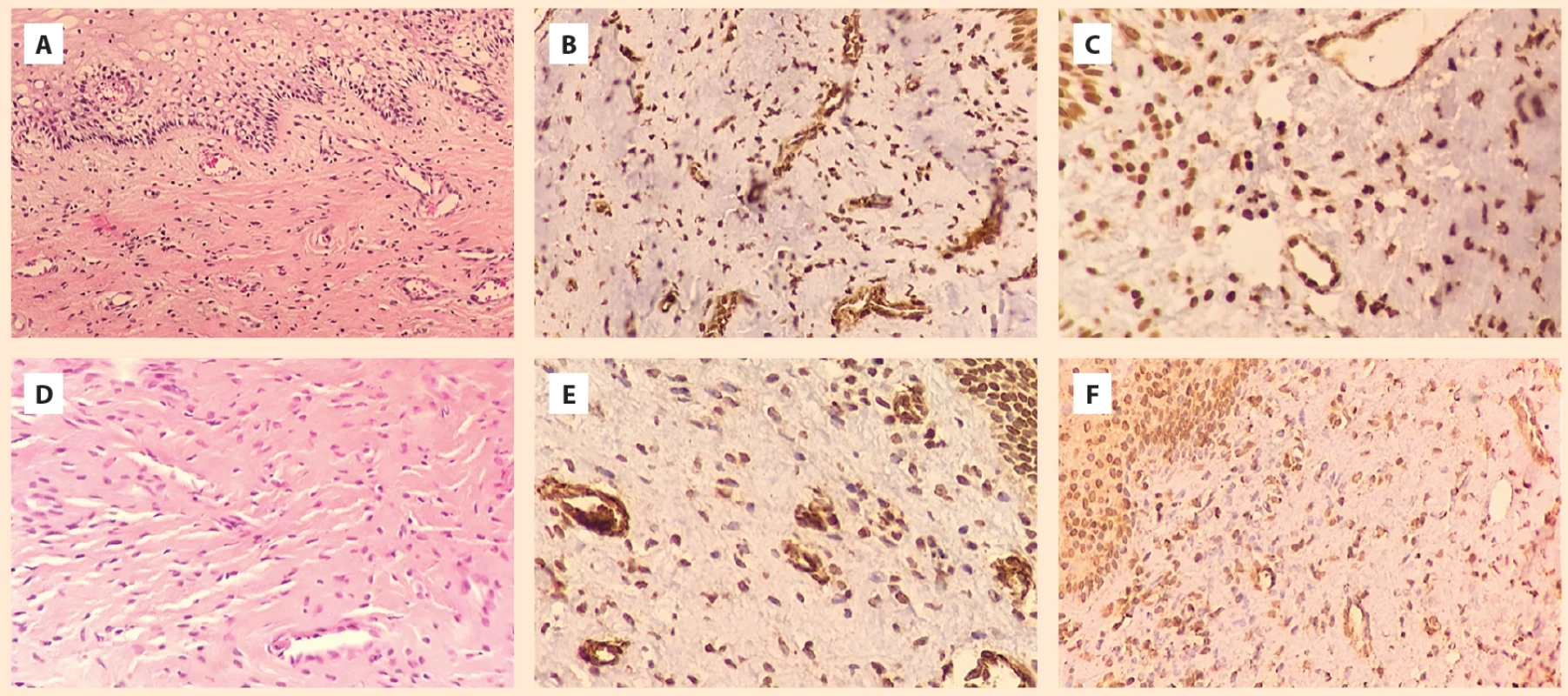
Figures shows the immunostaining of cases (Fig. 1) and controls (Fig. 2).
Statistical analysis
Data were analyzed by GraphPad Prism software. Results were expressed as medians, minimum and maximum values. The immunostaining of stromal markers was compared between groups using the Fisher’s exact test. The results of blood count parameters were compared between groups using the Mann-Whitney Test. Differences will be considered significant for P < 0.05.
In the immunohistochemical study, the agreement between two observers will be performed using kappa:
- - k < 0.4: weak agreement;
- - 0.4 ≤ k < 0.8: moderate agreement;
- - 0.8 ≤ k < 1.0: strong agreement;
- - k = 1.0: perfect agreement.
Results
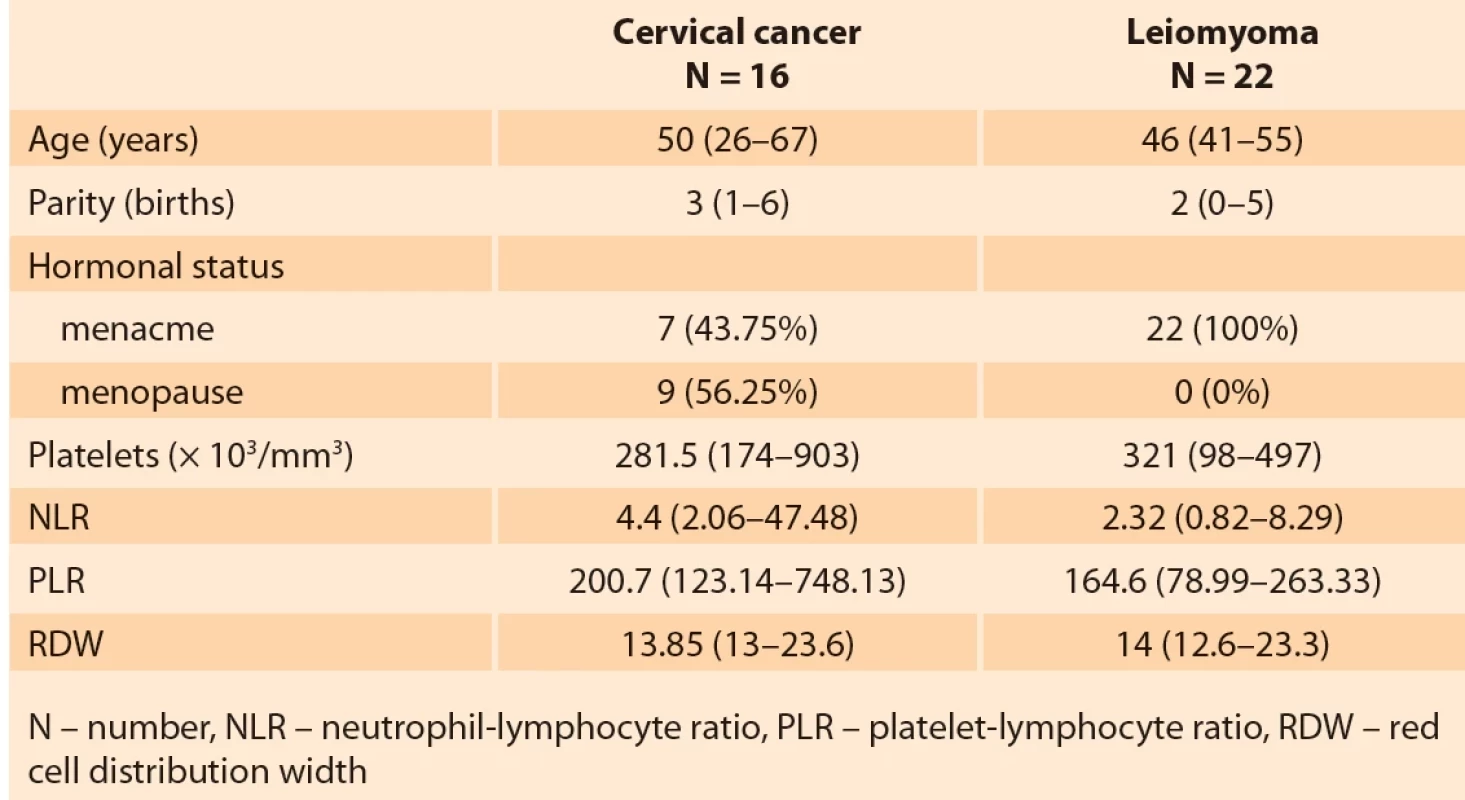
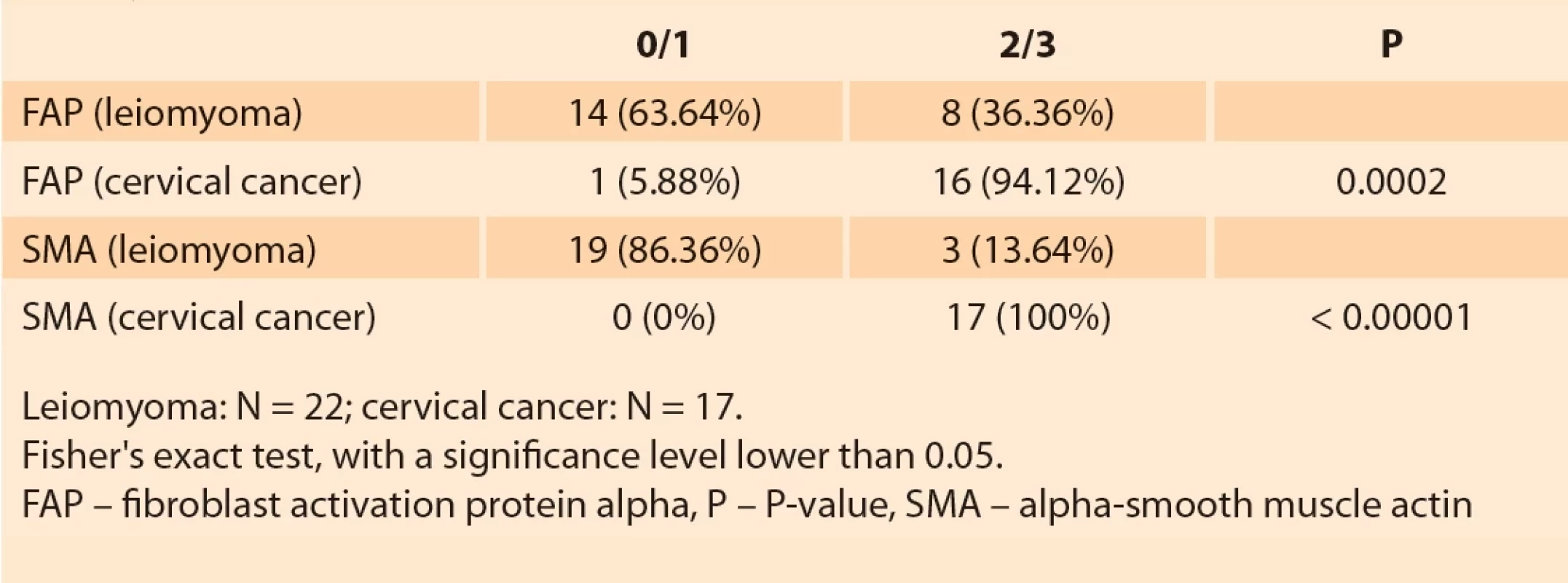
The general characteristics and parameters of the blood count of the groups of patients with cervical cancer and uterine leiomyoma are shown in Tab. 1. The histological type, histological grade and staging of the patients in the cervical cancer group are shown in Tab. 2.
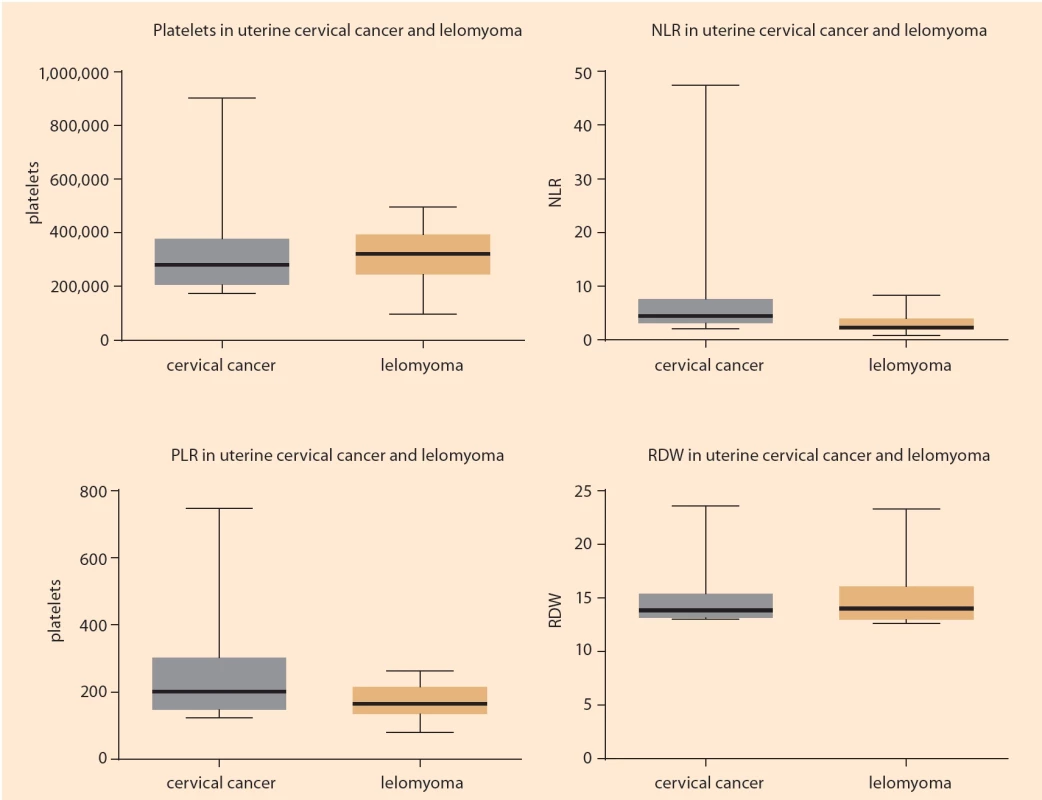
The NLR values were higher in the group of cancer patients (median = 4.4; minimum = 2.06; maximum = 47.48) compared to the control group (median = 2.32; minimum = 0.82; maximum = 8.29) (P = 0.0019). Regarding RPL values, these were also higher in the cancer group (median = 200.7; minimum = 123.14; maximum = 748.13) than in the leiomyoma group (median = 164.6; minimum = 78, 99; maximum = 263.33), but at the threshold of significance (P = 0.0675). There was no significant difference in platelet and RDW values when comparing these groups (P = 0.3986 and P = 0.9095, respectively) (Fig. 3).
Strong immunostaining (2/3) by FAP was more frequent in patients with cervical cancer when compared with patients with myoma (control group) (P = 0.0002). Regarding SMA, strong immunostaining (2/3) was also found more in the group of cancer patients compared to the group of patients with myoma (P < 0.00001) (Tab. 3).
NLR values were higher in the group of cancer patients (median = 4.4; minimum = 2.06; maximum = 47.48) compared to the control group (median = 2.32; minimum = 0.82; maximum = 8.29) (P = 0.0019). Regarding RPL values, these were also higher in the cancer group (median = 200.7; minimum = 123.14; maximum = 748.13) than in the leiomyoma group (median = 164.6; minimum = 78.99; maximum = 263.33), but at the threshold of significance (P = 0.0675). There was no significant difference in platelet and RDW values when comparing these groups (P = 0.3986 and P = 0.9095, respectively) (Fig. 3).
There was no significant difference in the evaluation of the two stromal markers in cervical cancer in the study of pathological prognostic factors (staging and histological grade) nor in the study of systemic factors (platelets, NLR, PLR and RDW).
Discussion
Tumor stroma contains distinct prognostic data evidenced by vascular cells and immune cells in the movement of neoplastic progression. FAP and SMA markers were evaluated in cervical cancer, and studies have been carried out showing that fibroblast behavior and characteristics influence therapeutic responses. The presence of immunostaining for SMA in the cancer cell is responsible for creating a permissive environment for tumor progression [7]. There is a strong relationship between the association of the transformation of cervical intraepithelial neoplasia into invasive carcinoma [8,9]. This reinforces our result, since in the present study, strong immunostaining for SMA was found more in the group of cancer patients when compared to the group of patients with myoma.
It is known that FAP has a potential therapeutic target, and its present labeling can be used for targeted therapy or imaging [10]. It is highly expressed in reactive stromal fibroblasts in epithelial carcinomas. FAP contributes to neovascularization within tumors, in addition to increasing tumorigenicity [11]. In this study, strong FAP immunostaining occurred more frequently in the group of patients with cervical cancer.
In addition to the degradation of the basement membrane, the invasion of cervical cancer is carried out by the remodeling of the interstitial stroma. Thus, some proteins and fibroblasts play an important role in the pathogenesis of cervical cancer [12]. FAP is an immunohistochemical marker with a potential therapeutic target in several types of gynecological and genitourinary carcinomas, but which did not show much prognostic significance in untreated cervical cancer, as its expression did not correlate with clinical outcomes. However, it has been shown that FAP may have value in targeted therapy of cervical neoplasia [10]. In our study, there was no significant difference in the assessment of the two stromal markers in cervical cancer in the study of pathological prognostic factors (staging and histological grade) or in the study of systemic factors (platelets, NLR, PLR and RDW).
The tumor microenvironment has been receiving more attention in recent years. Various inflammatory cells have a major role in the tumor environment [13]. Inflammation plays an important role in the initiation and progression of many cancers, including cervical cancer. Currently, the correlation between blood factors, easily identified by a blood count, and the prognosis and risk of dissemination in cervical cancer is known. The most studied are NLR, PLR, RDW and the isolated platelet count, which were used in this study. Research showed that in advanced cervical cancer higher values of NLR and PLR were found when compared to early stages [14]. High NLR is associated with lower overall survival and disease-free survival, and can be considered as an independent factor of worse prognosis [14] and is a biomarker to predict the development of cervical intraepithelial neoplasia [15]. Our study corroborates this information, since NLR values were higher in patients with malignant neoplasia than in the control group.
Regarding PLR, it is an independent risk factor for increased lymph node metastases, acting as a prognostic factor, especially if combined with NLR [16]. In the present study, higher values of PLR were also found in patients with neoplasms when compared to the control group, despite being at the limit of significance. Thus, the PLR together with high NLR seem to provide an increased risk for the development of cervical neoplasia. Platelet count and RDW were not relevant in the present study, although the increase in RDW in cervical cancer has already been described [17] including the consideration of thrombocytosis as a pre-treatment prognostic factor [18].
Some limitations also exist in our study. The main limitation is the small sample size of patients. However, despite the limitation, our study was the first in the literature to compare stromal markers and blood count parameters with a control group of patients with uterine leiomyoma, and may be a target for new studies related to carcinogenesis and new treatments for this disease, whose morbimortality still has so much impact, especially in developing countries. In addition, our results may have potential in future studies on the relationship between the stroma and systemic inflammatory response, bearing in mind that the blood count is a simple, inexpensive, and easy-to-collect test.
Conclusion
Strong immunostaining (2/3) by FAP and SMA were found more in the group of patients with cervical cancer when compared to the group of patients with myoma. The RNL values were higher in the cancer patient group when compared to the control group. Regarding RPL values, these were also higher in the cancer group than in the leiomyoma group, but at the threshold of significance. There was no significant difference in platelet and RDW values when comparing these groups. There was also no difference in the evaluation of the two stromal markers in cervical cancer in the study of pathological prognostic factors (staging and histological grade) nor in the study of systemic factors (platelets, NLR, PLR and RDW).
Sources
1. Li Q, Huang W, Zhou X. Expression of CD34, alpha-smooth muscle actin and transforming growth factor-beta1 in squamous intraepithelial lesions and squamous cell carcinoma of the cervix. J Int Med Res 2009; 37 (2): 446–454. doi: 10.1177/147323000903700220.
2. Menon H, Ramapriyan R, Cushman TR et al. Role of radiation therapy in modulation of the tumor stroma and microenvironment. Front Immunol 2019; 10 : 193. doi: 10.3389/fimmu.2019. 00193.
3. Zeltz C, Primac I, Erusappan P et al. Cancer-associated fibroblasts in desmoplastic tumors: emerging role of integrins. Semin Cancer Biol 2020; 62 : 166–181. doi: 10.1016/ j.semcancer.2019.08.004.
4. Da Silva AC, Jammal MP, Etchebehere RM et al. Role of alpha-smooth muscle actin and fibroblast activation protein alpha in ovarian neoplasms. Gynecol Obstet Invest 2018; 83 (4): 381–387. doi: 10.1159/000488088.
5. Da Silva AC, Jammal MP, Arvelos Crispim PC et al. The role of stroma in ovarian cancer. Immunol Invest 2020; 49 (4): 406–424. doi: 10.1080/08820139.2019.1658770.
6. Bhatla N, Aoki D, Sharma DN et al. Cancer of the cervix uteri. Int J Gynaecol Obstet 2018; 143 (Suppl 2): 22–36. doi: 10.1002/ijgo.12611.
7. Wu MP, Young MJ, Tzeng CC et al. A novel role of thrombospondin-1 in cervical carcinogenesis: inhibit stroma reaction by inhibiting activated fibroblasts from invading cancer. Carcinogenesis 2008; 29 (6): 1115–1123. doi: 10.1093/carcin/bgn077.
8. Li Q, Huang W, Zhou X. Expression of CD34, alpha-smooth muscle actin and transforming growth factor-beta1 in squamous intraepithelial lesions and squamous cell carcinoma of the cervix. J Int Med Res 2009; 37 (2): 446–454. doi: 10.1177/147323000903700220.
9. Xiao L, Zhu H, Shu J et al. Overexpression of TGF-b1 and SDF-1 in cervical cancer-associated fibroblasts promotes cell growth, invasion and migration. Arch Gynecol Obstet 2022; 305 (1): 179–192. doi: 10.1007/s00404-021-06137-0.
10. Sun L, Schroeder MC, Hagemann IS et al. Expression of potential biomarker targets by immunohistochemistry in cervical carcinomas. Int J Gynecol Pathol 2022; 41 (6): 628–635. doi: 10.1097/PGP.0000000000000853.
11. Lai D, Ma L, Wang F. Fibroblast activation protein regulates tumor-associated fibroblasts and epithelial ovarian cancer cells. Int J Oncol 2012; 41 (2): 541–550. doi: 10.3892/ijo.2012.1475.
12. Fullár A, Dudás J, Oláh L et al. Remodeling of extracellular matrix by normal and tumor--associated fibroblasts promotes cervical cancer progression. BMC Cancer 2015; 15 : 256. doi: 10.1186/s12885-015-1272-3.
13. Huang H, Liu Q, Zhu L et al. Prognostic value of preoperative systemic immune-inflammation index in patients with cervical cancer. Sci Rep 2019; 9 (1): 3284. doi: 10.1038/s41 598-019-39150-0.
14. Vaz de Lima PS, Silva Mantoani PT, Candido Murta EF et al. Laboratory parameters as predictors of prognosis in uterine cervical neoplasia. Eur J Obstet Gynecol Reprod Biol 2021; 256 : 391–396. doi: 10.1016/j.ejogrb.2020.11. 044.
15. Xu L, Song J. Elevated neutrophil-lymphocyte ratio can be a biomarker for predicting the development of cervical intraepithelial neoplasia. Medicine (Baltimore) 2021; 100 (28): e26335. doi: 10.1097/MD.0000000000026335.
16. Chen L, Zhang F, Sheng XG et al. Peripheral platelet/lymphocyte ratio predicts lymph node metastasis and acts as a superior prognostic factor for cervical cancer when combined with neutrophil: lymphocyte. Medicine (Baltimore) 2016; 9 (32): e4381. doi: 10.1097/MD.0000000000004381.
17. Kose M, Celik F, Kose SK et al. Could the platelet-to-lymphocyte ratio be a novel marker for predicting invasiveness of cervical pathologies? Asian Pac J Cancer Prev 2015; 16 (3): 923–926. doi: 10.7314/apjcp.2015.16.3.923.
18. Kozasa K, Mabuchi S, Komura N et al. Comparison of clinical utilities of the platelet count and platelet-lymphocyte ratio for predicting survival in patients with cervical cancer: a single institutional study and literature review. Oncotarget 2017; 8 (33): 55394–55404. doi: 10.18632/oncotarget.19560.
ORCID authors
S. Santana Otano 0000-0002-0763-4618
A. M. Melo 0000-0002-4578-4115
F. B. Ferreira Souza 0000-0001-6134-8973
J. Souto Morlin 0000-0002-6019-7170
E. de Castro Côbo 0000-0003-3020-2255
A. C. Macêdo Barcelos 0000-0001-5612-7009
A. Misson Rua Micheletti 0000-0003-2101-0366
M. Prata Jammal 0000-0003-2693-8817
E. F. Candido Murta 0000-0003-4014-1345
R. Simões Nomelin 0000-0001-6591-7619
Submitted/Doručeno: 31. 8. 2023
Accepted/Přijato: 20. 10. 2023
Prof. R. Simões Nomelini, MD
Laboratory of Applied Sciences for Women (LaCam)
Department of Gynecology and Obstetrics
Federal University of Triângulo Mineiro
Av. Getúlio Guaritá, s/n, Bairro Abadia
Uberaga-MG 38025-440, Brazil
rosekeila@terra.com.br
Labels
Paediatric gynaecology Gynaecology and obstetrics Reproduction medicineArticle was published in
Czech Gynaecology

2024 Issue 2
-
All articles in this issue
- Artificial activation of sperm motility in vitro
- Reactive oxygen species and their role in the andrological factor of couple fertility
- Metrics of uterine myomatosis symptomatology in clinical practice
- Trophoblast stem cells, trophoblast invasion, and organoids – advancements in gynecology
- Usage of the levonorgestrel releasing intrauterine system in perimenopause
- Combined contraceptives – the choice of estrogen matters
- Fecal incontinence risk factors and pregnancy
- Haemorrhagic stroke in pregnancy
- Interprofessional cooperation with a dermatovenerologist – specifics of hidradenitis suppurativa management in gynaecological practice
- New staging of endometrial carcinoma – FIGO 2023
- A new perspective on Endometrial Carcinoma classification and management strategies in context of molecular subtypes
- Dynamics of serum levels and reference ranges of copeptin in the 3rd trimester of pregnancy in healthy pregnant women with uncomplicated pregnancy and delivery
- Peritumoral stroma and systemic inflammatory response in cervical cancer
- Czech Gynaecology
- Journal archive
- Current issue
- About the journal
Most read in this issue
- New staging of endometrial carcinoma – FIGO 2023
- Usage of the levonorgestrel releasing intrauterine system in perimenopause
- Metrics of uterine myomatosis symptomatology in clinical practice
- Trophoblast stem cells, trophoblast invasion, and organoids – advancements in gynecology
